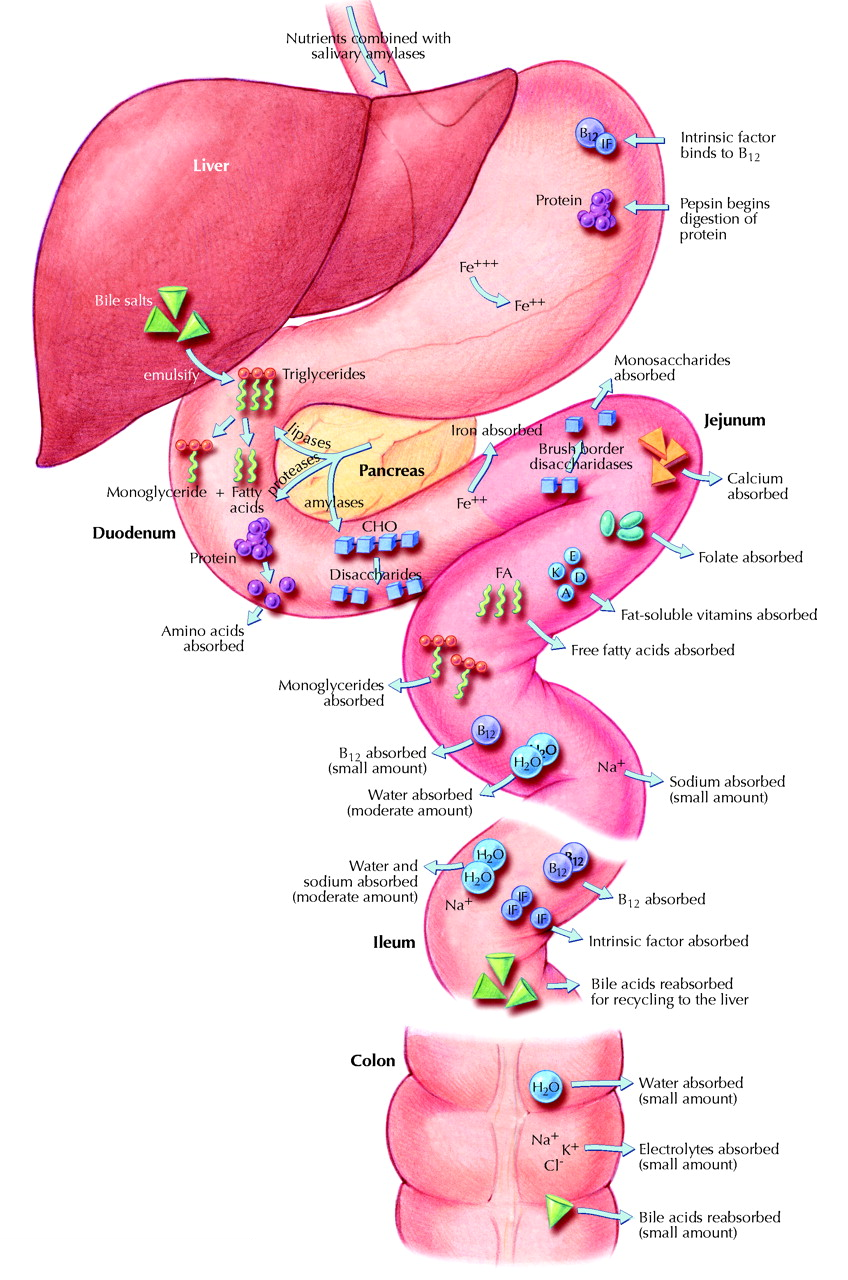
| In 1998 the growth of 47 children with inflammatory bowel disease was studied. The researchers concluded "Growth retardation is an important sign of chronic inflammatory bowel disease in prepubertal as well as adolescent children." (1) Moeeni and Day wrote an excellent review of the impact inflammatory bowel disease can have on growth in 2011. (2) In it they state, "In addition to presentation with loss of body weight or poor weight gains, children also commonly have altered patterns of linear growth." Small intestinal bacterial overgrowth, a manifestation of inflammatory bowel disease, has also been shown to lead to poor growth. (3) The mechanism of this lack of growth is clear. In order to grow a child's body needs building blocks to do so. If those building blocks are not absorbed properly from the gut then there is nothing the child's body can use to grow. On a more biochemical level, specific nutrients that contribute to optimal sleep, glucose metabolism, thyroid hormone function and other areas discussed here will not be absorbed in the presence of gastrointestinal dysfunction (image 1). |
The microbiome consists of trillions of microbial cells on and in the human body, the majority of the microbiome is found in the gut. The human microbiome also consists of the genes these cells harbor. Kunc, et al review very thoroughly how the microbiome in the gut can impact thyroid hormone, growth hormone and other hormones of the endocrine system in "Microbiome impact on metabolism and function of sex, thyroid, growth and parathyroid hormones". (5) Among many other things they discuss the role that lipopolysaccharides (LPS) have on the endocrine system. LPS is a toxic compound produced by gram-negative bacteria as part of their outer membrane. Elevated levels of LPS in the gut can have a number of negative effects on the endocrine system including thyroid and growth hormone, not to mention it's role in inflammation.
Children with Down syndrome experience a higher rate of and far too often under-recognized inflammatory bowel disease. The root of these gastrointestinal issues often lies in gut motility issues, untreated hypothyroidism, higher rate of c-section, higher rate of formula feeding and much higher rate of antibiotic use in infancy. The latter three lead to an imbalance in the gut microbiome in ways that extend into adulthood. (6)
| A healthy diet is the foundation to optimal health. Many parents work hard to get their children to eat vegetables, healthy fats, grass-fed meat, and little to no grains. While this may be an optimal diet for an adult it can have some consequences to growth in children. A diet that primarily consists of fats, meat and vegetables can lead to ketosis in a child, especially when mean chain triglycerides (MCT, coconut and palm oil) are also used heavily in the diet. Ketosis is a normal metabolic process that occurs in the body when there is not enough glucose present to produce energy. A ketogenic diet is popular for weight loss in adults because it is very effective for this purpose. Unfortunately, it leads to poor growth in children. (7,8,9) |
Children whose diet is high in sugar and grains, particularly refined grains like white bread, white crackers and white rice can experience decreased growth due to their diet lacking of nutrients and protein. Another means by which such a diet can impact growth is the negative effect it can have on the microbiome of the child which has far reaching effects on the child's overall health as well. A diet that is high in sugar and grains can lead to yeast overgrowth in the gut, especially in a child who has received antibiotics. Children who eat diets that are high in refined grains are typically low in fiber. Soluble fiber is one of the main food sources for healthy bacteria in the gut. Conlon and Bird have written a comprehensive review of the impact that diet can have on the gut microbiome and overall health in 2015. They stated, "Dietary means, particularly the use of a range of fibers, may be the best way of maintaining a healthy gut microbiota population." (12)
The way we breathe at night can have a big impact on the quality of our sleep. Mouth breathing at night can contribute to and even cause ADD/ADHD.(15) Mouth breathing actually changes facial structure in children as well which makes it even harder to nose breathe later in life. (16) Ensuring nasal passages are clear before going to bed either by blowing the nose, gently cleaning the nostrils with a moist q-tip, using saline nasal irrigation or spray can help increase the ability to nasal breathe. Enlarged adenoids can make nasal breathing difficult. Consult your child's doctor if you suspect enlarged adenoids which can lead to chronic ear infections, sinus infections and mouth breathing during the day.
Researchers in Sweden studied hormone levels in 37 snoring men and found that IGF-1 (a marker for growth hormone) increased after one month of using a nostril dilator to prevent snoring. About half of the men reported being less tired in the morning as well.(17) This study highlights the direct connection that breathing pattern and quality of sleep can have on growth.
Symptoms of poor sleep quality or not enough sleep in children are irritability, ADD/ADHD, behavioral issues, low energy, excessive yawning, falling asleep easily in the car and poor growth. Symptoms of sleep apnea include the same symptoms but might also include gasping and snoring at night that you would only hear if sleeping near your child. Sleep apnea can be silent in many children as well. If you're suspicious that your child may have sleep apnea it's best to speak with your doctor about getting a sleep study done. However, I have mixed feelings about sleep studies. On one hand, it's important to know whether a child is experiencing sleep apnea or not and on the other hand will it change any of the treatment that is already being taken to address underlying cause of suspected sleep apnea?
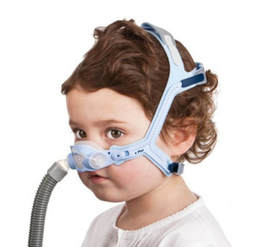
| The most commonly recommended treatment for sleep apnea next to tonsil and adenoid removal is CPAP (continuous positive airway pressure). I recommend avoiding CPAP machines for children who have sleep apnea. The mask has been shown to negatively impact facial development that can perpetuate and worsen the original cause of the sleep apnea, especially in children with Down syndrome who often have delayed growth of midfacial bones.(18) You can see from image 2 how wearing such a device that places constant pressure on the face for many hours at night would hinder proper anterior facial development. For my patients we address the root cause of the sleep apnea which will be addressed in a separate blog post due to the complexity of the topic. |
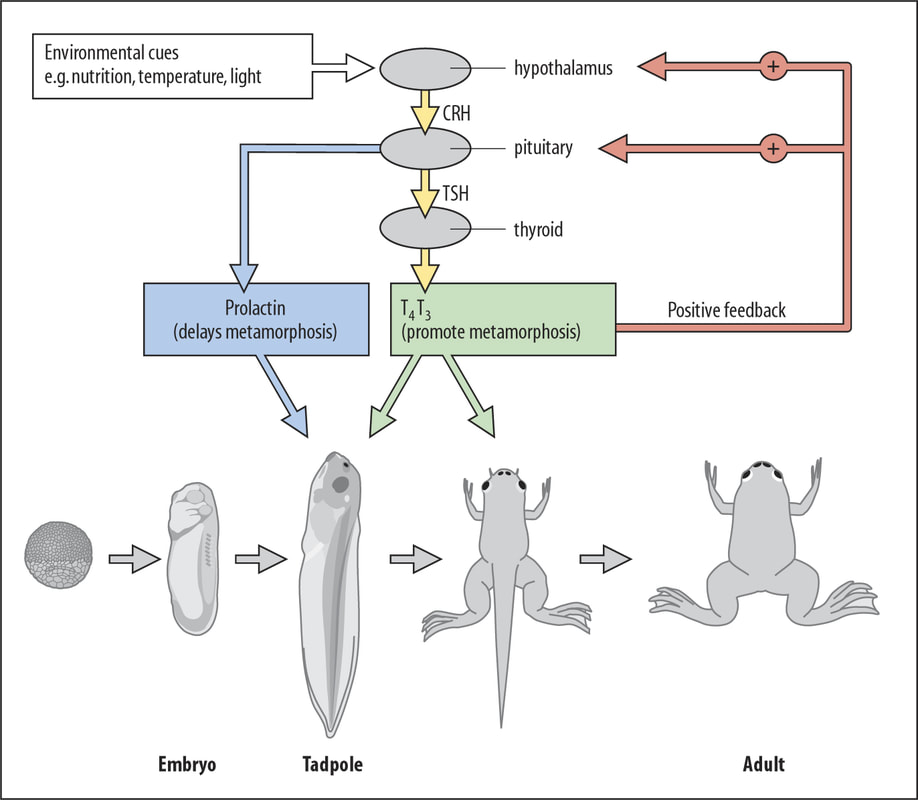
The cascade of events that occurs in order to get optimal levels of active T3 hormone within the cells of the body is complex. I review it briefly in my blog post "Pediatric Thyroid Reference Ranges". Many parents may believe that their child's thyroid hormone levels are normal or managed when in fact they are not optimal because the physician has not done complete thyroid labs which include TSH, free T4, free T3, reverse T3, TgAb and TPO. Management of hypothyroidism is complex and outside the scope of this blog post. For some patients thyroid hormone replacement therapy using natural desiccated thyroid hormone is warranted. We strive to treat the root cause of hypothyroidism in all of our patients if they are receiving hormone replacement therapy or not, because no one experiences just hypothyroidism. There is always an underlying cause that should not be missed.
| Many parents pursue the option of growth hormone injections for their child, even though this is the most invasive of all interventions that impact growth, without addressing the other underlying causes of growth delay. The screening test for checking growth hormone function is done through a routine blood draw. The labs ordered are IGF-1 and IGFBP-3. These are insulin-like growth factor 1 and insulin-like growth factor binding protein 3, respectively. If these are shown to be low the next step is to pursue a growth hormone stimulation test. This is a three to four hour test that is conducted in a hospital or clinic setting. The child receives an IV and an agent that is known to stimulate the release of growth hormone is administered through the IV. Blood samples are then drawn every 30 minutes to check if the child's pituitary has responded to injection of the stimulating agent. If it is shown that the child's pituitary response to the stimulating agent is low then the child qualifies for receiving daily injections of growth hormone. Some endocrinologists will prescribe growth hormone injections even if the child's pituitary does respond to the stimulating agent. This is known as passing the test. I warn against this, especially when addressing the other five factors has not been pursued. |
Several compounds have been studied for their effect on growth hormone secretion. These include glycine, taurine, glutamine, ornithine, vitamin B12, niacin and arginine. Arginine is actually one of the agents used in the growth hormone stimulation test due to it's well-known effect on increasing growth hormone secretion from the pituitary gland. Researchers in The Netherlands used a combination of glycine, glutamine and niacin in middle-aged and elderly subjects and found that it did increase GH secretion, but not IGF-1 levels. However, some subjects did experience increased IGF-1 levels and those subjects also experienced increased memory and vigour.(25) In 2014 a large group of international researchers came together to show the importance of taurine, whose synthesis is dependent on vitamin B12, in growth hormone regulation. They were able to show that taurine increased GH-dependent IGF1 synthesis in the liver. (26)
I want it noted that I'm not opposed to the use of growth hormone in children with Down syndrome when warranted. I simply want to ensure that all processes that impact growth are being explored and nothing gets missed in my patients.
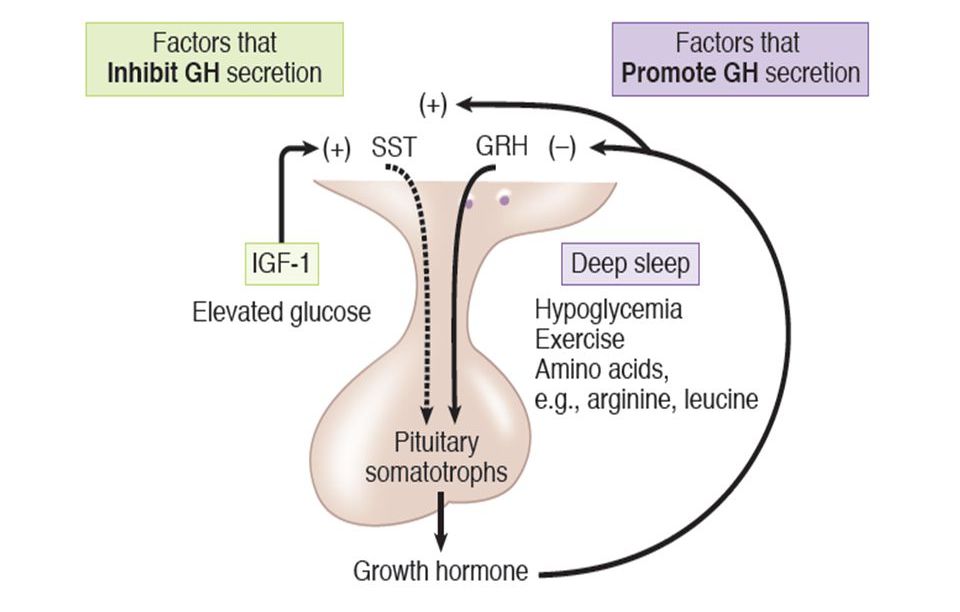
Growth hormone is well-known for it's positive impact on glucose metabolism. (28) It's role in glucose regulation is to raise blood sugar levels in times of hypoglycemia (low blood sugar). It does this by triggering gluconeogenesis in the liver. This is a normal process the body undergoes in the presence of hypoglycemia. Hyperglycemia (elevated blood sugar), therefore has the opposite effect. A negative feedback loop exists between blood sugar levels and the pituitary gland. The body adjusts growth hormone secretion based on it's need to tightly control blood sugar levels. Therefore, hyperglycemia leads to a decrease in growth hormone secretion. Other hormones involved in blood sugar control are glucagon, catecholamines and cortisol.
Thiamine and riboflavin deficiency can also lead to hyperglycemia.(32,33,34) These two B vitamins, along with alpha-lipoic acid are co-factors, meaning they are absolutely necessary, for the function of the enzyme pyruvate dehydrogenase. This enzyme shuttles the breakdown product of glucose (pyruvate) into the mitochondria where it can be used for energy. If this enzyme doesn't work the whole mechanism backs up and glucose levels increase in the blood. In my practice I pick up deficiencies of these two B vitamins often in my patients with Down syndrome. Deficiencies in these vitamins, sometimes severe, can be seen using an organic acid test. Not all patients with Down syndrome experience these deficiencies as they are more often seen in those who have malabsorption secondary to gut issues.
Bones are no exception when it comes to communicating with the nervous system. The skeletal system is very dynamic, especially in children. It's constantly receiving signals from the rest of the body to breakdown bone and build new bone in a process called remodeling and modeling. You can view the video below to learn more about this process.
A complete understanding of how the nervous system controls bone growth has yet to be developed by the scientific and medical community. However, some research does exist. Orthopedic physicians from Greece have written a review article outlining the role the peripheral nervous system plays in bone growth in children. In it they state, "Bone growth...has a very complicated regulation... Starting from the genetic factors, growth factor, insulin-like growth factor 1 (IGF-1), triiodothyronine, thyroxine, androgens, Indian hedgehog, fibroblast growth factors, bone morphogenetic proteins (BMPs), vascular endothelial growth factors (VEGF) are some of them which affect positively the bone growth." (36) They go on to discuss several other factors that implicate the nervous system in bone growth. One of these factors is CGRP (calcitonin-gene regulated peptide), one of several neuropeptides that was discovered only 30 years ago. It works to increase osteoblast activity. These are the cells of the bone that increase bone mineralization. CGRP is primarily released from sensory nerves and is a strong vasodilator.(37) CGRP also inhibits osteoclast (bone dissolving cells) activity.
Interestingly, CGRP levels were found to be low on blood spot tests of newborn infants with Down syndrome.(38) However, delayed bone growth and development in children with Down syndrome is clinically often recognized as simply due to them having Down syndrome. It's likely that supporting nervous system function in infants and children with Down syndrome can ultimately improve growth as well.
- Muscle weakness
- Cramps
- Muscle twitching
- Loss of muscle and bone
- Changes in skin, hair, or nails
- Numbness
- Loss of sensation or feeling in body parts
- Loss of balance or other functions as a side effect of the loss of feeling in the legs, arms, or other body parts
- Emotional disturbances
- Sleep disruptions
| What about the central nervous system and the brain? Do they play a role in bone growth? A team of physicians from the Univeristy of Iowa stated, "Disorders of nerves - central or peripheral - can have substantial influence on bone health and repair."(39) They shared images of electron micrographs taken in 1966 that clearly show myelinated and unmyelinated nerves present in cortical bone (image 6). This is important as the connection between the nervous system and bone is not often discussed in medical school or clinical practice. |
- Optimize glucose metabolism. This involves many factors and nutrients. Working with a functional medical or naturopathic physician who knows how to recognize when glucose metabolism is impaired is key. These physicians are trained in with how to test for and recognize nutrient deficiencies that contribute to and cause impaired glucose metabolism.
- Feed your child a whole foods diet that is high in organic vegetables, grass-fed meat, organic fruit, healthy fats and at least one serving of a whole grain per day. This whole grain should be gluten-free for those who are sensitive to gluten or have Celiac disease. One of my favorites is gluten-free oats.
- Avoid using too much coconut oil. No more than 1/2-1 tsp per day for most children.
- Optimize gut function by working with a knowledgeable physician who is trained to recognize and treat the root cause of gut dysfunction. A physician who uses organic acid testing and/or comprehensive stool analysis can help detect inflammation, SIBO, candida overgrowth, pancreatic dysfunction, fat malabsorption, leaky gut and more.
- Optimize sleep. This involves not only good sleep hygiene, but can also include tonsil and adenoid assessment, check of nutrient levels involved in calming the brain like zinc and magnesium, complete check of thyroid labs, resolving of gut issues, balancing of neurotransmitters, to name a few.
- Optimize thyroid hormone function by working with a physician who is knowledgeable about all of the causes of hypothyroidism and can prescribe the right form of thyroid hormone when needed.
- Screen for growth hormone deficiency with an IGF-1 and IGFBP-3 on your child's next routine blood draw.
- Do a thorough nervous system assessment with the help of your child's physician. If signs and symptoms of peripheral neuropathy exist the root cause should be ascertained and addressed as a means to optimize bone health and growth.
- Saha MT, Ruuska T, Laippala P, Lenko HL. Growth of prepubertal children with inflammatory bowel disease. Journal of Pediatric Gastroenterology and Nutrition. 1998;26(3):310–314.
- Moeeni V, Day AS. Impact of Inflammatory Bowel Disease upon Growth in Children and Adolescents. ISRN Pediatrics. 2011;2011:365712.
- Donowitz JR, Petri WA. Pediatric Small Intestinal Bacterial Overgrowth in Low-Income Countries. Trends in molecular medicine. 2015;21(1):6-15.
- DiBaise, JK. Nutritional Consequences of Small Intestinal Bacterial Overgrowth.Nutrition Issues in Gastroenterology. 2008.
- Kunc M, et al. Microbiome impact on metabolism and function of sex, thyroid, growth and parathyroid hormones. Acta Biochim Pol. 2016.
- Yasmin F, Tun HM, Konya TB, et al. Cesarean Section, Formula Feeding, and Infant Antibiotic Exposure: Separate and Combined Impacts on Gut Microbial Changes in Later Infancy. Frontiers in Pediatrics. 2017;5:200.
- Neal EG, Chaffe HM, Edwards N, Lawson MS, Schwartz RH, Cross JH. Growth of children on classical and medium-chain triglyceride ketogenic diets. Pediatrics. 2008;122:e334–40.
- Groleau V, Schall JI, Stakkings VA, Bergqvist CA. Long-term impact of the ketogenic diet on growth and resting energy expenditure in children with intractable epilepsy. Developmental medicine and child neurology. 2014;56(9):898-904.
- Vining E. P. G., Pyzik P., McGrogan J., et al. Growth of children on the ketogenic diet. Developmental Medicine and Child Neurology. 2002;44(12):796–802.
- Rogovik AL, Goldman RD. Ketogenic diet for treatment of epilepsy. Canadian Family Physician. 2010;56(6):540-542.
- Napoli E, Dueñas N, Giulivi C. Potential Therapeutic Use of the Ketogenic Diet in Autism Spectrum Disorders. Frontiers in Pediatrics. 2014;2:69.
- Conlon MA, Bird AR. The Impact of Diet and Lifestyle on Gut Microbiota and Human Health. Nutrients. 2015;7(1):17-44.
- Peirano PD, Algarín CR, Chamorro RA, et al. Sleep alterations and iron deficiency anemia in infancy. Sleep medicine. 2010;11(7):637-642.
- Dosman C, Witmans M, Zwaigenbaum L. Iron’s role in paediatric restless legs syndrome – a review. Paediatrics & Child Health. 2012;17(4):193-197.
- Um YH, Hong S-C, Jeong J-H. Sleep Problems as Predictors in Attention-Deficit Hyperactivity Disorder: Causal Mechanisms, Consequences and Treatment. Clinical Psychopharmacology and Neuroscience. 2017;15(1):9-18.
- Harari D, Redlich M, Miri S, Hamud T, Gross M. The effect of mouth breathing versus nasal breathing on dentofacial and craniofacial development in orthodontic patients. Laryngoscope. 2010;120((10)):2089–93.
- Löth S, et al. Improved nasal breathing in snorers increases nocturnal growth hormone secretion and serum concentrations of insulin-like growth factor 1 subsequently. Rhinology. 1998 Dec;36(4):179-83.
- Roberts SD, Kapadia H, Greenlee G, Chen ML. Midfacial and Dental Changes Associated with Nasal Positive Airway Pressure in Children with Obstructive Sleep Apnea and Craniofacial Conditions. Journal of Clinical Sleep Medicine : JCSM : Official Publication of the American Academy of Sleep Medicine. 2016;12(4):469-475.
- Gilbert SF. Developmental Biology. 6th edition. Sunderland (MA): Sinauer Associates; 2000. Metamorphosis: The Hormonal Reactivation of Development.
- Bucholz DR. More similar than you think: Frog metamorphosis as a model of human perinatal endocrinology. Dev Biol. 2015 Dec 15;408(2):188-95.
- Castells S, Beaulieu I, Torrado C, Wisniewski KE, Zarny S, Gelato MC. Hypothalamic versus pituitary dysfunction in Down's syndrome as cause of growth retardation. J Intellect Disabil Res. 1996 Dec;40(Pt 6):509–517.
- Chaplin JE, Kriström B, Jonsson B, Tuvemo T, Albertsson-Wikland K. Growth hormone treatment improves cognitive function in short children with growth hormone deficiency. Horm Res Paediatr. 2015
- Myrelid A, Bergman S, Elfvik SM, Jonsson B, Nyberg F, Gustafsson J, et al. Late effects of early growth hormone treatment in down syndrome. Acta Paediatr. 2010;99:763–9.
- Anneren G, Tuvemo T, Carlsson-Skwirut C, et al. Growth hormone treatment in young children with Down’s syndrome: effects on growth and psychomotor development. Archives of Disease in Childhood. 1999;80(4):334-338.
- Arwert LI, Deijen JB, Drent ML. Effects of an oral mixture containing glycine, glutamine and niacin on memory, GH and IGF-I secretion in middle-aged and elderly subjects. Nutr Neurosci. 2003 Oct;6(5):269-75.
- Roman-Garcia P, Quiros-Gonzalez I, Mottram L, et al. Vitamin B12–dependent taurine synthesis regulates growth and bone mass. The Journal of Clinical Investigation. 2014;124(7):2988-3002.
- Berg JM, Tymoczko JL, Stryer L. Biochemistry. 5th edition. New York: W H Freeman; 2002. Section 30.2, Each Organ Has a Unique Metabolic Profile.
- Kim S-H, Park M-J. Effects of growth hormone on glucose metabolism and insulin resistance in human. Annals of Pediatric Endocrinology & Metabolism. 2017;22(3):145-152.
- Hua Y, Clark S, Ren J, Sreejayan N. Molecular Mechanisms of Chromium in Alleviating Insulin Resistance. The Journal of Nutritional Biochemistry. 2012;23(4):313-319.
- Chutia H, Lynrah KG. Association of Serum Magnesium Deficiency with Insulin Resistance in Type 2 Diabetes Mellitus. Journal of Laboratory Physicians. 2015;7(2):75-78.
- Mooren F.C., Krüger K., Völker K., Golf S.W., Wadepuhl M., Kraus A. Oral magnesium supplementation reduces insulin resistance in non-diabetic subjects—A double-blind, placebo-controlled, randomized trial. Diabetes Obes. Metab. 2011;13:281–284.
- Thakur K, Tomar SK, Singh AK, Mandal S, Arora S. Riboflavin and health: a review of recent human research. Crit Rev Food Sci Nutr. 2017;57(17):3650–3660.
- Alam MM, Iqbal S, Naseem I. Ameliorative effect of riboflavin on hyperglycemia, oxidative stress and DNA damage in type-2 diabetic mice: Mechanistic and therapeutic strategies. Arch. Biochem. Biophys. 2015;584:10–19.
- Alaei Shahmiri F, Soares MJ, Zhao Y, Sherriff J. High-dose thiamine supplementation improves glucose tolerance in hyperglycemic individuals: a randomized, double-blind cross-over trial. Eur. J. Nutr. 2013;52(7):1821–1824.
- Niedzwiedzki T., Filipowska J. Bone remodeling in the context of cellular and systemic regulation: the role of osteocytes and the nervous system. J. Mol. Endocrinol. 2015;55:R23–36.
- Gkiatas I, Papadopoulos D, Pakos EE, Kostas-Agnantis I, Gelalis I, Vekris M and Korompilias A (2017) The Multifactorial Role of Peripheral Nervous System in Bone Growth. Front. Phys. 5:44.
- Russell FA, King R, Smillie S-J, Kodji X, Brain SD. Calcitonin Gene-Related Peptide: Physiology and Pathophysiology. Physiological Reviews. 2014;94(4):1099-1142.
- Nelson PG, et al. Selected neurotrophins, neuropeptides, and cytokines: developmental trajectory and concentrations in neonatal blood of children with autism or Down syndrome. Int. J. Dev. Neurosci. 2006;24:73–80.
- Jones KB, Mollano AV, Morcuende JA, Cooper RR, Saltzman CL. Bone and Brain: A Review of Neural, Hormonal, and Musculoskeletal Connections. The Iowa Orthopaedic Journal. 2004;24:123-132.
- Hammond N, Wang Y, Dimachkie M, Barohn R. Nutritional Neuropathies. Neurologic clinics. 2013;31(2):477-489.




 RSS Feed
RSS Feed